In analogy to its twin physical property known as "superconductivity", the term "superfluidity" was originally coined to describe the frictionless flow of liquid helium which could be observed under some circumstances at temperatures below 2.17 K. Today, it is understood as a collective name for all the unusual properties of the quantum liquid phases of both stable isotopes of helium (3He and 4He) and their mixture. Superfluidity have also been observed in very sparse laser-cooled clouds of other atoms that undergo Bose-Einstein condensation.

To understand superfluidity of helium, we must first note the differences between the two isotopes. While the nucleus of the abundant 4He consists of two protons and two neutrons, has an integral spin and consequently is a bosonic particle, the rare isotope 3He, missing one neutron, has a half-integral spin and thus is fermionic.
While both isotopes behave quite similarly at room temperature (differing only in their density, diffusivity, and possible nuclear reactions), they obey different quantum-mechanical statistics: the Bose-Einstein distribution applies to the bosonic 4He, while the fermionic 3He obeys the Pauli exclusion principle and follows the Fermi-Dirac distribution. In consequence, they exhibit remarkably different properties when cooled down, their transitions to superfluid phases occur at temperatures differing by a factor of 1000 and via different mechanisms.
From the phase diagram of 4He we see that unlike other substances, it has no triple point, in fact, no solid-gas equilibrium line at all. Indeed, if cooled down at atmospheric pressure, helium would not freeze even at absolute zero, and to produce solid 4He, an external pressure of at least 25 bar must be applied.
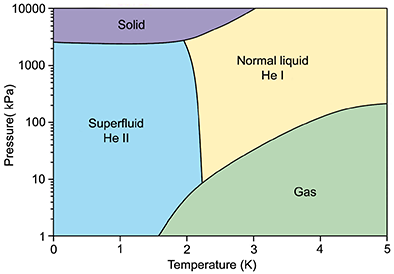
Instead, liquid 4He forms a new phase upon crossing the so-called "lambda line" roughly at 2.2 K, which intersects the saturated vapour line at the "lambda-point", 2.17 K. This new phase was historically called He II to distinguish it from normal liquid 4He (called He I) and is of prime interest to us, because it is the one exhibiting superfluidity.
Amazingly, He II gives us the opportunity to visually observe direct consequences of quantum physics on macroscopic scales. Naturally, these were the first of its interesting properties discovered and studied.
When the second order phase transition from He I to He II is observed in a glass cryostat, it is immediately apparent that the pool boiling of He I is suddenly suppressed. The reason is that the heat conductivity of He II is at least 3 million times higher than that of He I, and is sufficient to suppress temperature gradients favouring the existence of preferred bubble nucleation sites. Instead, He II is cooled only via surface evaporation. Remarkably, He II will adhere to almost any material it is in contact with, creating a nanoscopic layer on its surface. This superfluid film will cause flow over the walls of any open vessel in contact with He II, unless prevented artificially.
More extraordinary properties of He II have been observed in experiments that involved heating a volume of fluid separated from the rest of He II by tightly compressed powder or other realisation of what is now known as a "superleak" - an arrangement of microscopic solid particles that represents a block for classical fluids, but through which superfluid He II flows easily. It was demonstrated that in such an arrangement, a temperature increase caused by heating will also result in an overpressure that will propel the fluid contained upward in a fountain jet. This phenomenon is called the thermo-mechanical effect or simply "fountain effect". An opposite effect exists in He II as well, it is known as the mechano-caloric effect.
The most successful phenomenological model of He II is called the two-fluid model. While the first ideas were reported by L. Tisza in 1938, the model was reformulated and completed by L. Landau in 1941. The model is based on the assumption that the flow of He II can be decomposed into the flow of two interpenetrating fluids, the normal fluid and superfluid components. While the normal component is viscous and carries the entire He II entropy, the superfluid component is an inviscid fluid, capable of frictionless flow. Such an approach enabled researchers to clarify various experimental observations. For example, the abnormally large heat conductivity of He II can be explained as the occurence of a thermally driven flow of the normal component without any net mass flow (i.e., the superfluid component flows in the opposite direction).
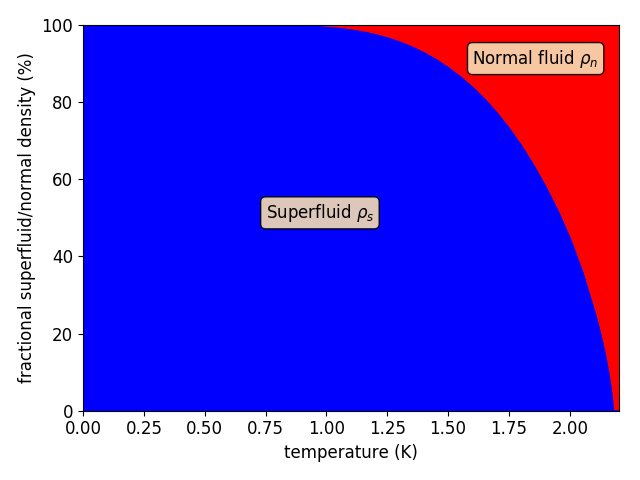
The atoms of the superfluid component of He II display, to a certain extent, collective behaviour. To decribe such state of matter, we can define some order parameter, a complex function of space and time that determines the local density of He atoms and represent the entire volume of the superfluid component as one wave of matter. Aplying the laws of quantum mechanics, one can derive that such an object must be irrotational, i.e., the vorticity (curl of velocity of the superfluid component) must be zero in every point in space. However, early experimental observations of rotating He II, which mimics the rotation of a solid body, showed that the flow of the superfluid component can be of non-zero vorticity. In consequence, in 1955, R. Feynman introduced one-dimensional topological defects that occur in the superfluid component, called quantized vortices, to explain the experimental findings.
The vortices have a few angstrom in size and their cores do not contain the superfluid component. But most importantly, the circulation (a closed-loop integral of the superfluid component velocity) of the vortices is constrained to be quantized in units of the quantum of circulation. Since the energy of the vortex depends quadratically on its quantum number, multiply-quantized vortices are energetically unfavourable and only singly-quantized vortices exist in the superfluid component of He II.
The phase diagram shows us that superfluid 3He can exist in several different phases at temperatures below its critical temperature (2 mK), three orders of magnitude lower than that of 4He, explaining why superfluidity in 3He was discovered much later (the 70's).
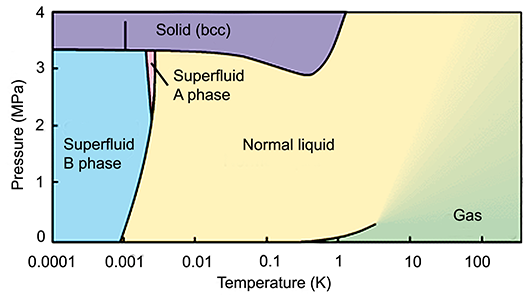
Having a non-zero nuclear spin, 3He nuclei also have a corresponding magnetic moment and both normal and superfluid 3He thus are magnetic liquids. Unlike Cooper pairs in conventional superconductors, 3He pairs have a total spin S equal to 1. Depending on the external magnetic field, the preferred quantum mechanical states the pairs occupy may change, resulting in not one, but three different superfluid phases of 3He with different magnetic order.
In zero or low magnetic fields (and low temperature, pressure), the pairs may occupy states with all three possible values of projected spin Sz = -1, 0, 1; forming the B phase with an isotropic energy gap. At higher magnetic field (or temperature, pressure) the A phase is formed and the pairs occupy only the states with Sz = -1 or 1. Due to the resulting magnetic order, the A phase has a highly anisotropic energy gap with two nodes at the poles. A third phase called A1 exists as well (at very high magnetic fields and pressures, and temperature very close to the critical one), where all the pairs have their spins aligned with the external magnetic field, Sz = 1. Yet more superfluid 3He phases exist in the cores of quantized vortices or in aerogel.
At high temperatures, the two isotopes have very similar electrophysical properties and are therefore infinitely soluble in each other. If a mixture containing mostly 4He is cooled down below the lambda point or lower (depending on the concentration of 3He), it will eventually reach a transition to a superfluid phase. In this phase, the superfluidity occurs in 4He and 3He acts approximately as an additional normal component.
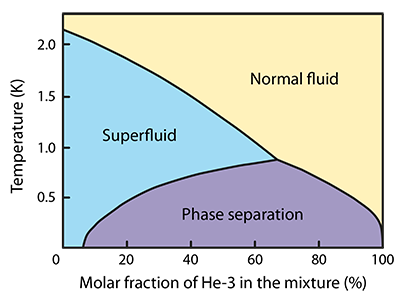
At temperatures below roughly 0.8 K, the infinite solubility of the two isotopes breaks down due to quantum effects and a phase separation occurs - the mixture splits into two separate phases, the 3He-rich phase floating atop the dilute phase. In the zero temperature limit, 4He is no longer soluble in 3He, but a finite solubility of 3He in 4He remains, up to around 6% concentration.
This allows forcing a flow of 3He through the interface of the two phases, which, if applied in the direction from the 3He rich phase to the dilute one, results in local cooling of the mixture and forms the basis of operation of dilution refrigerators - powerful cooling devices capable of reaching and maintaining temperatures as low as 2 mK continuously.